CRISPR-based gene therapies have yet to be approved by the FDA, despite their relative affordability and ease when compared to traditional gene therapies. This may change in 2023, as CRISPR Therapeutics and Vertex Pharmaceuticals announced that their biologics licensing applications (BLAs) to the U.S. Food and Drug Administration (FDA) were completed, including a request for priority review, which would shorten the FDA’s traditional twelve-month review of the application to eight months. This timeline opens the possibility for the first CRISPR gene-edited therapy to be available for interstate commerce within the year.
Background
CRISPR/Cas9 complexes were initially discovered in the natural immune systems of bacteria to protect them from viral invaders. The CRISPR component is a sequence complementary to a specific “target” sequence in a patient’s genome. It is sometimes referred to as the Guide RNA, as it guides the entire complex to the place within the genome where editing will occur. The Cas9 enzyme is the protein commonly depicted as a pair of metaphorical scissors, as it cuts DNA to allow for the insertion/deletion of intended genetic material. In the medical field, CRISPR genetic editing can be harnessed to potentially edit the genomes of individuals affected by currently incurable genetic diseases.
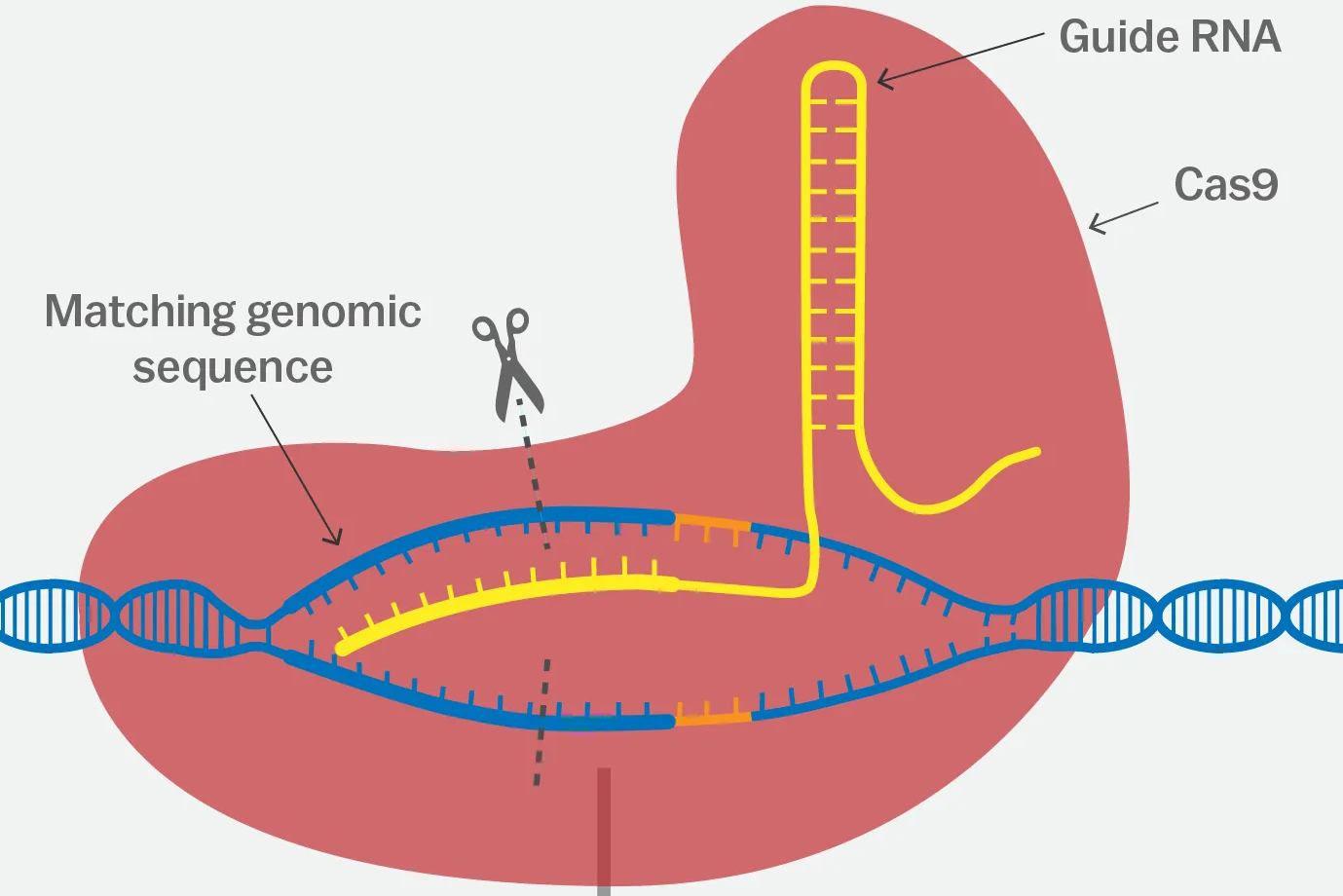
CRISPR/Cas9 is much more accessible than other FDA-approved gene therapies, due to its relative affordability and ease of use. Yet the FDA has shown caution when it comes to its approval. Major limitations of CRISPR-based medicines include:
- The potential for off-targeting, wherein the complex incorrectly recognizes and binds to a sequence similar to the target sequence.
- The triggering of the body’s immune response by CRISPR/Cas9, as it originates from bacteria.
- The multi-faceted ethical concerns that come with genetic editing.
Methods and results
Despite concerns, researchers with CRISPR Therapeutics and Vertex Pharmaceuticals are in the final stages of clinical trials and are up for FDA approval with their CRISPR/Cas9 therapy for genetic blood disorders, called exagamglogene autotemcel (exa-cel).
Patients with sickle cell disease (SCD) and transfusion-dependent beta-thalassemia (TDT) who are participating in these trials have stem cells collected from their own blood. These cells are then edited with CRISPR/Cas9 outside of the body. Once the edited cells are introduced back into the body the patients are treated in accordance with traditional hematopoietic stem cell transplant (HSCT) procedures to establish high levels of fetal hemoglobin (HbF) production. HbF is the protein that carries oxygen throughout the bloodstream during fetal development.
The addition of HbF to a patient with SCD allows for a reduction or potential elimination of vaso-occlusive crises, wherein sickled red blood cells block blood flow to specific tissues, depriving them of oxygen and triggering an extremely painful immune response.
Within patients with TDT, increased levels of HbF reduce or eliminate the life-long dependence on blood transfusions that come with the characteristic severe anemia of the disease.
CRISPR Therapeutics and Vertex Pharmaceuticals are in stage III of clinical trials, assessing both adults and children with SCD/TDT. They presented the adult data from 75 patients (31 with SCD and 44 with TDT) at the European Hematology Association Congress in December 2022.
All of the 31 patients with severe SCD that had been experiencing recurrent vaso-occlusive crises saw an elimination of the crises at their follow-up after exa-cel infusion (follow-up ranging from 2.0 to 32.3 months).
Of the 44 blood-transfusion-dependent patients with TDT, 42 were transfusion-free after exa-cel infusion (follow-up ranging from 1.2 to 37.2 months) and two were at the 75% and 85% marks in transfusion-reduction.
These CRISPR-based therapies show solid potential to change the idea of “incurable” blood diseases.
This research supports the biologics licensing applications (BLA) of CRISPR Therapeutics and Vertex Pharmaceuticals. A BLA is a request to the FDA to introduce a biological product, in this case the exa-cel gene therapy, to the interstate market. Within the BLA, there is a request for Priority Review, which would shorten the FDA’s traditional twelve-month review of the application to eight months, potentially allowing for the first CRISPR gene therapy to be FDA-approved within 2023.
Although the exa-cel CRISPR gene therapy is not approved just yet, it is an exciting innovation for CRISPR research and patients affected by “incurable” genetic diseases.
References
- Frangoul, H., Altshuler, D., Cappellini, M. D., Chen, Y.-S., Domm, J., Eustace, B. K., Foell, J., de la Fuente, J., Grupp, S., Handgretinger, R., Ho, T. W., Kattamis, A., Kernytsky, A., Lekstrom-Himes, J., Li, A. M., Locatelli, F., Mapara, M. Y., de Montalembert, M., Rondelli, D., … Corbacioglu, S. (2021). CRISPR-Cas9 gene editing for sickle cell disease and β-thalassemia. New England Journal of Medicine, 384(3), 252–260. https://doi.org/10.1056/nejmoa2031054
- Kingwell, K. (2023, April 3). First CRISPR therapy seeks landmark approval. Nature News. https://www.nature.com/articles/d41573-023-00050-8
- Ran, F. A., Hsu, P. D., Wright, J., Agarwala, V., Scott, D. A., & Zhang, F. (2013). Genome engineering using the CRISPR-cas9 system. Nature Protocols, 8(11), 2281–2308. https://doi.org/10.1038/nprot.2013.143
- Vertex and CRISPR therapeutics complete submission of rolling biologics license applications (Blas) to the US FDA for exa-Cel for the treatment of sickle cell disease and transfusion-dependent beta thalassemia. Vertex Pharmaceuticals. (2023, April 3). https://investors.vrtx.com/news-releases/news-release-details/vertex-and-crispr-therapeutics-complete-submission-rolling
- Vertex and CRISPR therapeutics present new data on more patients with longer follow-up treated with exagamglogene autotemcel (exa-cel) at the 2022 European Hematology Association (EHA) Congress. Vertex Pharmaceuticals. (2022, June 11). https://investors.vrtx.com/news-releases/news-release-details/vertex-and-crispr-therapeutics-present-new-data-more-patients
Jordan is a Chemical Engineering student and undergraduate researcher at the University of Florida. She is passionate about genetic engineering and science communication. Her goal is to portray scientific information in a way that is understandable and beneficial to everyone, whether they are immersed in the scientific community or not.

2 replies on “First CRISPR-based Gene Therapy Could be Approved in 2023”
Cool article
I agree